Like most songbirds, American Redstarts stake out and defend territories during the breeding season. But winter territories are also defended vigorously by individual redstarts, although winter territories of female American Redstarts sometimes overlap with those of their neighbors. Redstarts also tend to return to the same winter territories year after year.
Brown-headed Cowbirds, which lay their eggs in the nests of other birds, often try to parasitize American Redstart nests. Redstarts are known to respond aggressively to a cowbird near their nest, or even abandon a nest containing cowbird eggs, though occasionally they will accept cowbird eggs and raise cowbird young.
On this page
Description of the American Redstart
BREEDING MALE
The American Redstart has a black-tipped tail with brightly colored patches at the base which are visible when the tail is fanned, something redstarts do often.
Males have a black head, throat, and upperparts, with orange patches on the sides, wings, and tail.
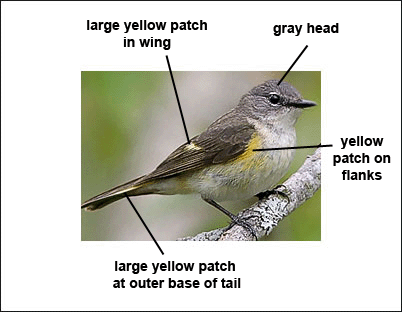
Female
Females have a gray head, throat, and upperparts, with yellow patches on the sides, wings, and tail.
Seasonal change in appearance
None.
Juvenile
Similar to adult females, but duller.
Habitat
American Redstarts inhabit second growth woods and tree-lined stream banks.
Diet
American Redstarts eat insects, and some seeds and berries.
Behavior
American Redstarts forage actively by flycatching or hovering.

Range
American Redstarts breed from northwestern Canada south across the northwestern U.S. and the eastern half of the U.S. They winter from Mexico south to South America. The population is stable.
More information:
Bent Life History
Visit the Bent Life History for extensive additional information on the American Redstart.
Wing Shape
The shape of a bird’s wing is often an indication of its habits and behavior. Fast flying birds have long, pointed wings. Soaring birds have long, broad wings. Different songbirds will have a slightly different wing shape. Some species look so much alike (Empidonax flycatchers) that scientists sometimes use the length of specific feathers to confirm a species’ identification.
Fun Facts
Male American Redstarts often have 2 or 3 mates.
American Redstarts are often very tame and curious.
Vocalizations
The song is a series of sharp notes. A squeaky flight call is also given.
Similar Species
Bay-breasted Warbler
The Bay-breasted Warbler has a mix of red and black colors but is not easily mistaken for the redstart.
The yellow or orange wing and tail patches, together with the tail fanning habit, make redstarts easy to identify.
Nesting
The American Redstart’s nest is a cup of grasses, rootlets, and lichens and is lined with finer materials. It is placed in the fork of a tree, often quite low.
Number: 3-4.
Color: whitish in color with darker markings.
Incubation and fledging:
The young hatch at about 12 days and fledge at about 9 days, though remaining dependent on the adults for some time.
Bent Life History of the American Redstart
Published by the Smithsonian Institution between the 1920s and the 1950s, the Bent life history series of monographs provide an often colorful description of the birds of North America. Arthur Cleveland Bent was the lead author for the series. The Bent series is a great resource and often includes quotes from early American Ornithologists, including Audubon, Townsend, Wilson, Sutton and many others.
Bent Life History for the American Redstart – the common name and sub-species reflect the nomenclature in use at the time the description was written.
SETOPHAGA RUTICILLA RUTICILLA (Linnaeus)
HABITSCONTRIBUTED BY ALFRED OTTO GROSS
The southern redstart is one of the commonest warblers in New England, perhaps second in abundance only to the yellow warbler. It is a resident of our forests, but unlike the ground-inhabiting ovenbird it is strictly an arboreal species and is much more frequently seen. Because of the brilliant, contrasting colors of the male and its extreme vivaciousness it is better known, and to many it has proven to be a favorite warbler. The bright flashes of orange-red which it proudly displays in its frequently spread tail and fluttering wings has suggested to the imaginative Cubans the beautiful name Candelita, the “little torch,” which in such unusual numbers brightens the dark shadows of their tropical forests. In the Province of Quebec, the Canadian French know it as “La Fauvette ~ Queue Rousse,” another allusion to the bright red patches in the male’s tail. Like many other American birds the name redstart was given it by the earlier settlers who bestowed upon it the name of a familiar Old World form which also has an orange-red tail but is otherwise very different in coloration and belongs to another family.
One of the most pleasant experiences I have had with warblers in the Maine woods, occurred on a bright morning in May when I suddenly came upon three male redstarts. One of them was working along a horizontal birch limb with its wings lowered in characteristic fashion and eagerly scanning every twig in its quest of insects. The other two were whirling about, darting upward, floating downward displaying their gorgeous colors, then snapping up their victims as they dashed again and again through a haze of midges hovering in the sunshine of the clearing. This scene, as it was enacted against a contrasting background of rich green hemlocks and firs, always comes to my mind when I think of redstarts.
The redstart’s manner of catching insects, as well as the shape of its bill and the well-formed rictal bristles, suggests the flycatchers; but instead of perching and patiently waiting for his prey to come near, as does a true flycatcher, the redstart is continually in action, dashing here and there after flying insects or perhaps snapping up larva dangling in midair at the end of its long silken fiber.
Spring: The redstart, which spends the winter in the West Indies and Central and South America, makes its first appearance in spring on the Florida Peninsula and coasts of the other Gulf States during the first week of April. By the middle of the month the vanguard reaches North Carolina, Kentucky, and Missouri. During the last week of April they progress as far as Maryland, Pennsylvania, and Indiana. We may expect the first arrivals in New England, New York, Ohio, Michigan, and Iowa during the first week of May. By the middle of the month they reach Quebec, Ontario, and Manitoba; and before the end of May they arrive at the outposts of their summer range in northern British Columbia.
After the redstarts leave their winter quarters early in April, they cross the Gulf of Mexico, following no particular route, or proceed from the West Indies via Florida. They thus enter the United States on a wide front extending westward a distance of some 2,000 miles to Texas. From this wide area of entry they spread out fanlike, as do members of many other species, until they reach across the widest part of the North American Continent from Newfoundland on the east to British Columbia on the Pacific. The migration from the winter quarters to the extreme northwestern section of its nesting range requires somewhat less than two months.
Milton B. Trautman (1940) from many years of observations at Buckeye Lake, Ohio, finds that the first redstarts arrive there in April. A marked increase in numbers occurs during the first week of May, the period of maximum abundance extending from May 10 to May 20. A marked decrease occurs after May 22, and a few days later virtually all that remain are summer residents. This condition seems to be typical throughout this belt of the redstart’s range. At points farther south or north the dates would be correspondingly earlier and later.
Courtship: The males are the first to appear and can be heard singing at the time of their arrival. The females come a few days or a week later. Although the first redstart arrivals in Maine may be expected early in May, it is several weeks before the bulk of the resident birds are to be found on their breeding grounds. The males exhibit unusual aggressiveness and indulge in a good deal of fighting in defending their chosen territories. Sometimes two males may be seen hovering in flight as they peck viciously at each other and then perhaps dart into cover with their tails spread and their whole being in a quivering belligerent state. Again, the strutting antics of the amorous males may be seen as they display before the females.
Joseph J. Hickey (1940) as a result of a very intensive and thorough study of the redstarts on a 40-acre area in Westchester County, N. Y., gives us an excellent account of their courtship and territorial aspects. The redstart, he says: Is a highly territorial species. Males advertised their presence by their typical well-known song and by formalized territorial displays that apparently served to define boundaries and reduce fighting. These displays consisted of short, horizontal, semicircular flights made with stiffened wings and out-spread tails. These performances were frequently observed between males, less commonly between females and never between. a male and a female where a question solely of territory was involved. Hingston’s interpretation of the function of warning coloration in plumages seemed to be particularly applicable in these cases. Low, repeated quit, quit notes could be heard when the displays were concluded and the birds returned to their perches. As far as could be observed, the same performances seemed to serve as some part of the male’s courtship of tbe females. On all exciting occasions, of course, both sexes spread their tails like many other wood warbiers. Flight songs appeared to be absent. Singing perches, if present, were largely undetected by the observer. One male which took up territory in a blackberry-locust association sang frequently on April 30 and May 8 only one to two feet from the ground. Three males were once watched for an entire morning before females had arrived in the area: one was quite obviously patrolling the boundaries of his territory, the two others seemed to be moving back and forth on an indefinite and irregular axis, which approximated the length of their territories.
A special effort was made In 1937 to learn the number of unmated males. Twenty-four occupied rigidly fixed territories on the study area. Twenty of these birds were definitely mated. Of the remaining four, one held a territory until at least June 13, another until at least June 19, a third until June 20, and a fourth until June 26. If all four, or even half of these were unmated, the percentage of paired birds would he between 83 and 91. * * * An indeterminate number of unmated wandering males also exist, A male in the immature plumage spent the entire morning softly singing and gradually working its way along 800 yards at the top of the ridge. This bird was furiously driven off by males and females whenever It passed through their territories. Plumage notes on 48 males on territory showed that only four (8.3 per cent) were in immature plumage. All four were paired and possessed territories of the same size as those of the adult males.
The size of territories was about one acre or less, but in one Instance was compressed to about half an acre. Approximately twenty-two pairs (or males) each year occupied the 39.93 acres under investigation. Their boundaries were observed in two cases to break down on June 17, when young were being fed in the nest. Interspecies competition or jealously were seldom In evidence. Redstarts and Ovenbirds were the two most dominant species of the slope and both would sing in the same tree without the slightest evidence of hostility. The former was once seen briefly fighting with a Black and White Warbler.
Nesting: In Maine the redstart is an inhabitant of the hardwood or mixed deciduous and coniferous woodlands. These may be in low, damp situations but many of them are found in the second growth of trees and brush of our dry sandy plains. Often the nests are found in the thick growth of small trees which border the forest of larger trees, as well as in places where trees have sprung up along the roadsides. Others may be found in alder and willow thickets bordering our streams and ponds. In New York, it nests in low, damp woods; it has been found in mixed woodland with a considerable growth of pine and hemlock; and in the Adirondacks its nesting site is often in places where spruces predominate. In Ohio, Michigan, and other sections of the Middle West it frequents the maple, elm, ash, and pin-oak association of the larger, more mature swamp forests, although sometimes found among similar trees and brush in the larger upland woods. In the far Northwest it shows a decided preference for willow trees and alder thickets.
There is a tendency for the redstart to forsake the seclusion of the woodlands and, like the yellow warbler, to build its nest in trees and shrubbery adjacent to human habitations. It may even use the exterior of a human dwelling for a nesting site. Annie Lyman Sears of Waltham, Mass., reports that a nest was built on a bracket above a Venetian ironwork lantern hanging before the front door of her home The redstart has also taken up its residence in the parks located in the midst of some of our densely populated cities.
The most usual site of the nest of the redstart is an upright, 3- or 4-pronged crotch of a dead or live hardwood sapling, such as a maple, elm, ash, or birch. The majority of the nests range from 4 to 20 feet above the ground with an average height of about 7 feet. However, in many instances the redstart has selected other than typical nesting sites.
J. Claire Wood (1904) reports finding a nest in Wayne County, Mich., that was partly sunken into the ground at the base of a gooseberry bush between two logs, a very unusual situation. Verdi Burtcli has sent us a photograph of a redstart’s nest, found near Brancliport, N. Y., May 31, 1929, that was built in the forks of a bush only a few inches above the ground. Richard C. Barlow reported that he found a nest at Tabusintac, New Brunswick, June 26, 1919, that was 20 inches above the ground; it straddled and was near the tip of the horizontal limb of a spruce standing on the edge of a bog in rather open woods. On June 25, 1927, at Squam Lake, N. H., Arthur C. Bent found a nest 5 feet up in the fork of a dead branch that had fallen and was leaning upright against a striped maple sapling. At Brunswick, Maine, I discovered a nest held by a thick cluster of branches growing upward from the side of the main trunk of a large elm tree more than 2 feet in diameter at the base. This nest was not less than 30 feet from the ground. Nests placed in crotches formed by limbs branching from the main trunk of large trees are not rare. J. Claire Wood (1904) also reports an extreme case of a redstart’s nest built 70 feet above the ground in a large oak tree.
The redstart may also choose a small shrub or bush for its nesting place. A. C. Bent found a nest with three eggs, at Squam Lake, N. H., built in a small mountain laurel among a large clump growing in deep woods. The nest was not over 2 feet above the ground, a situation often selected by the black-throated blue warbler.
In June 1942 a pair of redstarts built their nest in a lilac shrub next to a house on Boody Street, Brunswick, Maine. It was saddled in a cluster of branches about 5 feet above the ground and was only a foot from a front window. Pedestrians frequently passing by on a walk only a few feet distant did not seem to disturb the birds in the least, and the birds even allowed me to take motion pictures of them as I stood only 4 feet away, completely exposed to view. During the same month another pair occupied a mockorange bush growing in the yard of a home only a short distance from this nest. Their nest rested on a horizontal branch about an inch in diameter and was securely anchored by three small upright branches. M. B. Trautman (1940) found six nests of the redstart in the Buckeye Lake region, Ohio, all in grapevine tangles 10 to 20 feet above the ground. J. Claire Wood (1904) describes an interesting nest in a grapevine in Wayne County, Mich., as follows “This vine reached downward about ten feet from the first limb of a large oak tree and thence upward to within a fodt of the starting point, forming a swing, and at the bottom of the loop the nest was placed. It was a windy day and the nest swung over a space of five feet, but madam clung to her treasures perfectly unconcerned.”
Another curious departure from the usual nesting habits of the redstart is its use of old or deserted new nests of other species such as the vireos. On June ~, 1898, in Yates County, N. Y., Verdi Burtch (1898) found a red-eyed vireo’s nest which had been newly lined with red bark fibers that the redstart usually uses to line its nests in that locality. The nest contained three fresh eggs of the redstart. Others have come to my attention: Three in which red-eyed vireo’s nests were used, one in which a yellow-throated vireo’s nest was used, and another in which a nest started by a yellow warbler was used. Since these instances were in widely separated places, it indicates the practice is not isolated or merely local. The nest is built entirely by the female, which exhibits more than ordinary architectural ability. It is a firm, compact structure composed of various plant and bark fibers, small rootlets, and flexible grass stems. The outside walls are covered with plant fibers, ornamented with small lichens, bit.s of birch bark, bud scales, seed pods, and vegetable down firmly bound in with a liberal use of spider web. The interior is lined with fine grasses, weed and bark fibers, and often with the hair of horse or deer.
Feathers of various kinds are frequently used, apparently as an added decoration. These may be ordinary small chicken feathers, but sometimes those of highly colored birds such as the tanager or indigo bunting are chosen. Ernest H. Short (1893) gives an account of a redstart’s nest made up almost entirely of the feathers of a wood thrush that had been killed by an owl near the nesting site.
Each nesting season I put out masses of cotton and tow for various birds to use as nesting material. A redstart which built its nest in a tall, slender maple nearby not only used some of the long delicate fibers of tow but studded the whole structure of the nest with small tufts of cotton, giving it a most unusual appearance, not unlike many similarly decorated yellow warbler’s nests I have seen. The nest of the redstart somewhat resembles that of the yellow warbler but it is a neater structure with thinner walls, especially at the rim. It is higher than wide and in this respect differs from the goldfinch’s nest, which is wider than high.
The average dimensions of a series of nests are as follows: outside diameter 27/s inches, inside diameter 1½ inches; outside depth 3 inches, and inside depth of the cup 1½ inches. The chief departure from these dimensions are of nests that are built in very narrow crotches where the upright supporting branches are close together at their point of origin. In such cases the birds continue to build until a sufficient height is reached to give a satisfactory width. The height of such nests may exceed 5 inches and are acutely V-shaped in section. High nests also occur when the redstart builds over cowbird’s eggs deposited before its own eggs have been laid.
It requires a week or ten days for the industrious female to build the nest. The task is performed in a most expert manner. The outside framework is first constructed, and then she enters the nest to adjust the lining, turning around and around and pressing her breast against the sides until the desired symmetry is attained. The final bits are dextrously manipulated by the bird’s bill and seem to be pasted on, according to various observers, with the aid of her own saliva.
Louis Sturm (1945) observed the building of one nest, which he states was completed in the course of 2% to 3 days. He estimated that the bird made 650 to 700 trips in building the nest. The first egg in this case was laid 2 days after the completion of the nest.
William Brewster (1936) presents a detailed account of the building of a redstart nest that is interesting and informative:
At about S A. M. on May 17th a female redstart brought a long, transparent, silky-looking fiber: apparently that of a milkweed stalk: to a gray birch in front of the cabin and, placing it at the intersection of a rather stout branch with the main stem, began moving it about until its position suited her, when she pressed it down firmly by rubbing it with the side of her head which she turned slowly from side to side. This was literally the first bit of nest material that was put into the crotch. Many others of an apparently similar kind were brought during the forenoon and treated In the same way, although the bird had to use her bill rather vigorously In tamping some of the more refractory ones into place. She worked busily and steadily until noon when the foundation of the nest was finished.
At about 2: 30 P. M. she began the frame by attaching one end of a strand of fibrous material to the right hand side of the trunk a little above the branch on which the foundation was laid and fastening the other end to the foundation on the same side, the strand inclining downward at an angle of about 30~ or 4Q0ï Next, another strand was placed on the left side in the same position as the first, the ends of the two overlapping on the trunk. Then a third piece was brought and one end rubbed lightly against the center of the strand on the right, the opposite end being carried a little beyond that of the left hand strand. Next a fourth straud was rubbed on the trunk a little above the upper end of the left hand guy, to the middle of which the opposite was fastened by rubbing the two together. Each piece of fiber was fashioned into the general shape of the nest as soon as it was attached to both ends and more were brought and carried from point to point until a complete framework of about the size and shape of the half of a hen’s egg was erected around and resting on the foundations. This framework was so delicate that it looked as if the merest breath of wind would blow it away. During its construction, the bird worked entirely from the outside, standing on the branch and shaping each piece of fiber with her head.
The next day (May 18th) she began using cocoon fibers as well as milkweed hark. The former she obtained from a groove near the top of the cabin door in front of which she would hover on rapidly vibrating wings until the exertion compelled her to alight for a moment to rest and regain breath, either on top otrthe door or its hand knob. As soon as she had filled her bill, she would fly to the birch, alight on the branch and distribute her load around the inside of the nest; then, hopping into it and squatting down with her head and tail raised and back deeply hollowed, she would move slowly around to the right and then to the left, making usually a half but sometimes a whole turn and, with her head and breast, pressing the materials which she had brought into the meshes of the framework until they were completely filled. Working thus, always from the inside, she modeled and remodeled until by the constant application of fresh material she had transformed the original skeleton framework into a compact, firmly-woven nest. Occasionally she would drop directly into the nest without first alighting outside but she did not attempt to perch on its rim until it was nearly completed.
After putting in the lining, which consisted of horse hair, dry grass, and shreds of birch, grapevine and mullein bark, she drew the loose ends which had been left projecting or hanging down around the outside of the nest. These ends were drawn and tucked In to bind the lining and were held down by the bird’s breast until all within her reach were secured. She then managed to rub them still more firmly into place by craning her head over the rim of the nest and bringing her bill, throat and neck to bear on its top and inner and outer surfaces at one and the same time.
After this nest was finished, it was frequently visited by Black and White Creepers, Yellow Warblers and Red-eyed Vireos, all of whom attempted to appropriate some of its component materials for their own domiciles. They sometimes succeeded in getting away with a few strands despite the vigilance of the Redstart, who defended her castle with the greatest spirit.
Eggs: The usual number of eggs in a complete set of the redstart 15 4 but the total ranges from 2 to 5 (I have never found a set containing more than 4 eggs); this number seems to vary in different sections of its range. J. Claire Wood (1904) reported that out of 143 nests with eggs 9 of them had 5 eggs; other observers have reported finding 5 eggs, but sets of more than 4 eggs are not common. Although the first set generally consists of 4 eggs, if these are destroyed the second set seldom exceeds 3 eggs and very often there may be but 2.
The eggs are subject to considerable variation in their size and markings; the average short diameter of a series of 50 eggs is 12.3 millimeters and the average long diameter 16.2 millimeters. They are ovate but some tend toward short ovate. They have a slightly glossy texture with a ground color that varies from white to creamy-white, greenish white, or grayish white. The eggs are speckled, spotted, and frequently blotched with various shades of brown with undertones of gray and drab. There is generally a concentration of markings at the larger end, forming a distinct wreath. The amount and intensity of the markings vary; some eggs may be almost free of markings, on others the markings are so dense as to nearly conceal the ground color.
Incubation: The period of incubation is 12 days, and is performed entirely by the female. Normally only one brood is reared during any one season.
B. W. Baker (1944) in 3 hours of observation at a nest during the morning noted that the female left the nest seven times, the length of her absence varying from 2 to 10 minutes, while the time spent on the nest was 144 minutes or 80 percent of the observation time. In nests that I have had under observation in Maine, the male was seen to deliver food to the female at frequent intervals and perhaps for this reason these females left the nest less often.
A. D. DuBois writes to me of an interesting reaction of a nesting female to a mirror: “The spunky little female flew at a small mirror which I held over the nest on the end of a stick; she snapped her bill and even grew bold enough to peck at it. On June 3 she again fought the small mirror as I held it up to look into the nest, but she returned to her eggs immediately when I withdrew the obnoxious lookingglass.
Young: In one nest under observation at Brunswick, Maine, the first egg hatched while the adults were away from the nest. The male was the first to return to greet the youngster and his first act after an inspection of the nest was to remove the eggshells. The male also delivered the first food, a small minced larva, when the young was about 2 hours old. When the female returned, which was shortly after the male’s first visit, she carefully inspected the nest, then immediately settled down to brood the young and the three eggs. The male never brooded but he was very attentive in feeding the young. If the female was at the nest when he arrived, the food was presented to her and she in turn fed it to the young, but if she was away the male fed the young directly. On the first day the youngster was brooded 75 percent of the time. There were three young on the following day, and the fourth egg proved to be sterile. The amount of time required for brooding was more or less dependent on weather conditions. On a cool, rainy day more time was spent on the nest than when a moderate temperature prevailed. When it rained the female extended her wings over the edge of the nest and her head was directed upward toward the oncoming rain drops. She thus provided an effective roof to shield the helpless young from a disastrous soaking. The brooding instinct is very strong and if the young were removed from the nest for purposes of study and photography, the female mechanically brooded the empty nest the entire time the young were out, seemingly unaware of their absence. In the afternoon the nest was exposed to the direct. rays of the sun and on hot days the female perched on the edge of the nest with her wings half spread and her back toward the sun. At such times she panted incessantly in order to control her temperature through her intricate air-sac system. It is just as necessary for the adult to protect the young from, heat as it is to keep them warm on cool days. The amount of time spent in brooding becomes much reduced as the young grow older, and by the time they have attained their covering of feathers and acquire a temperature control there is little need for brooding.
The male, if it is an old bird, usually exhibits greater wariness in approaching the nest than does the female. Miss Cordelia Stanwood has found the reverse to be true of one first-year male, which seemed to show no fear at all of her presence at the nest, whereas the female hesitated to feed the young at such times. Perhaps the older bird’s wariness is due to more experience in life.
The part played by the two sexes in feeding the young varies with different individuals; usually both take an equal part, but at times either the male or the female may take the major responsibility. Miss Stanwood studied a nest of the redstart in which the female had disappeared, apparently killed. In this case the male took over all of the arduous duties of feeding the family and of caring for them after they left the nest. Since the young are fed 8 to 10 times every hour it takes the resourcefulness of both parents, let alone one, when a mate is lost.
At the time of hatching, the young have their eyes closed and are naked except for limited tufts of down found on the dorsal tracts including the crown. By the third day the eyes show slightly through very narrow slits and the papillae of the wing feathers make their appearance. By the sixth day the tips of the feathers are unsheathed; from this time on the young preen their feathers a great deal and they may be seen picking at the growing feathers, an action that apparently accelerates the unsheathing process. By the eighth day the juvenal plumage is well established, and the young may leave the nest but if not disturbed or excited usually remain a day longer.
Bernard W. Baker (1944) found the weight of a young on the day of hatching was 1.1 gin. and at 7 days the same young had increased its weight to 7.5 gin. The tarsus of the same bird was 6 mm. in length on the first day and 17 mm. on the seventh day.
As with other warblers, the nest is kept scrupulously clean not only of vermin hut of the feces, which are anticipated and disposed of as soon as they appear. During the first two days the fecal sacs are swallowed by the adults, but as the young grow older, more and more of them are carried away and dropped at some distance from the nest. This task, like that of feeding the young, is performed by both parents.
J. Claire Wood (1904) presents an interesting account of a nest of seven eggs which was shared by two pairs of birds, as follows: “When found one female was upon the nest and the other perched close beside it. They were equal]y demonstrative of anxiety as I ascended the tree. The eggs were in two layers and slightly incubated. Being of two distinct types there was no difficulty in separating them into sets of three and four. This was not a case of polygamy, as both males were present. All four were living in perfect harmony and understanding, which is remarkable from the fact that the males are inclined to pugnacity and firmly attack all intruders of their kind that invade their chosen territory.” Whether or not polygamy ever exists among redstarts, as with the ovenbird, to my knowledge has never been, determined.
Dr. A. F. Skutch describes an injury feigning performance of a pair of adult redstarts which he observed at Ithaca, N. Y., on June 13, 1931. These birds apparently had young in the vicinity. He writes: “While passing through a tract of low, swampy woods, I was led by the excited chirping of a pair of American redstarts to the discovery of their nest in the crotch of an elderberry bush about 9 feet above the ground. When I attempted to learn by feeling with my finger-tips what it contained, the birds gave such a demonstration as I have seldom witnessed. The female approached the closer to me, advancing within easy reach as she chirped her distressed reproaches; but the male, although he remained at a somewhat greater distance, was more active in his efforts to lead me away. Although I felt that to them it was a most anxious occasion, I confess that I deliberately prol6nged their agony that I might delight my eyes with the gorgeous display of the male redstart as he perched on a long twig with tail spread and wings fully extended, vibrating: it seemed to me: as rapidly as a hummingbird’s. What a splendid color-contrast in the full black and the orange-salmon of the wings flashing into a blur, while the rich colors of the relatively motionless tail showed so clearly! Descending to the broad surface of a skunk-cabbage leaf, he continued his manifestations of distress: but how can so airy and refulgent a creature convincingly express despair? Next, falling to the ground, he danced over it with spread tail and fluttering wings; but it impressed me rather as some fairy dance than a wounded bird attempting to avert grave danger from his nest. The female redstart’s display was similar but less intense, while her paler colors made it less spectacular. When next I returned my attention to the nest, I found it empty. Since it is inconceivab]e that the birds should have become so excited over an empty nest, I had no doubt that fledglings were hiding close by, possibly having scrambled to safety while my delighted eyes were fastened upon the parents. The attendants of tender nestlings in a nearby nest made no such demonstration upon my approach.”
Adults feeding young out of the nest are commonly seen throughout the month of July. There is a tendency for them to remain in the vicinit.y of the nesting site. but they are less noisy than the young of other species and hence are much less in evidence.
Plumages: The plumage of the redstart is distinctive and can be easily distinguished from those of other warblers. The young males, even those in the first nuptial plumage, are similar to the females; and, hence, the identification of the sexes of the younger individuals is more difficult.
The natal plumage is hair-brown and is present as small tufts located on the crown and various tracts of the dorsal part of the body.
The juvenal plumage is well established at the time the young leave the nest and in the course of a few weeks is completed with all of the feathers unsheathed. Jonathan Dwight, Jr. (1900), has described the juvenal plumage as follows: “Above, including the sides of the head, deep sepia-brown. Wings and tail deep olive-brown, the basal portion of the primaries, secondaries and outer rectrices pale lemon-yellow, the secondaries and tertiaries edged with dull olive-green, the coverts with wood-brown paler at the tips. Below, pale primrose-yellow, hair brown on the chin, throat and breast. Bill and feet dusky pinkish buff darkening to brownish black when older.”
George A. Petrides (194Th) studied the plumage of a young redstart which he kept in captivity after it had left the nest. He found that at 10 days of age the tail feathers began breaking out of their sheaths; at 13 days the yellow areas on the wings first appeared and by 20 days the yellow patches of the tail became completely exposed. It was not until the young were 26 days old that the yellow patches of the wings were fully visible.
When the bird was 22 days old the slate-gray juvenal plumage of the occipital and dorsal tracts was being replaced in quantity by the olive-green first winter plumage. The yellow underwing coverts also began to appear at this time; previously the underwing areas had been naked. The postjuvenal molt was thus begun before the juvenal plumage was fully acquired.
The first winter plumage involves changes in the body feathers and wing coverts but not the rest of the wings or the tail. Dwight (1900) describes this plumage as follows:
Unlike the previous plumage. Above, the pileum, nape and sides of neck mouse.gray, the back olive-green, often tinged with brownish orange, the upper tall coverts clove-brown. The wing coverts become dull olive-green. Below, dull white, ashy and pinkish buff suffusing the chin and throat, and orangeochraceons or deep chrome-yellow area on either side of the breast, the color tinging the breast and sides. Orbital ring, white.
First nuptial plumage acquired by a partial prenuptial moult, which involves chiefly the head and throat, where a few black feathers In patches are acquired. A few may be found scattered sparingly elsewhere and new white feathers on the chin are the rule. Abrasion and fading make birds paler above and whiter below. In this species, which is unique among our warbiers during the first breeding season in wearing an immature dress strikingly different from the adult, the renewal Is reduced to a minimum.
Adult winter plumage acquired by a complete postnuptlal moult in July. The black and orange-red dress is assumed, the black feathers often having a faint huffy edging. SometImes the orange basal part of the primaries or of the rectrices fails to develop and yellow, as in the first winter, takes its place.
Adult nuptial plumage acquird by wear. The abrasion of the black plumage is in places so slight that there might be some replacement by new feathers, but it is not apparent. Fading is not obvious, except of the flight feathers.
The plumages and moults [of the female] correspond to those of the male. First differs in first winter plumage which is browner, the breast patches merely yellow tinged and the basal part of the rectrices much paler yellow, this color usually absent from the base of the primaries and reduced in extent on the secondaries. Some specimens are much like males. The first nuptial plumage is acquired by a very ilmited, sometimes suppressed prenuptial moult. The adult winter plumage is scarcely different from the first winter, a little grayer on the back and the yellow area on the wings greater. The adult nuptial plumage is apparently the previous plumage plus wear.
Pure and partial albinistic and melanistic forms of the redstart have been reported.
Thos. D. Burleigh (1944a) describes a hybrid between the redstart (Setopkaga r-aticilla) and the parula warbler (Composotkiypis americana) which he secured on Cat Island, Miss., 9 miles offshore from Gulfport:
This specimen bears a superficial resemblance to the female Redstart hut close scrutiny reveals marked differences. The bill Is not broadly wedge-shaped as in the Redstart, and the rictal bristles are developed only to the same degree as in the Parula Warbler, in this respect differing markedly from the genus Setophaga. The wings have the middle and greater wing-coverts broadly tipped with dull white, forming two distinct bands, and there is a complete absence of the speculum on the inner remiges. The tail, although noticeably longer than in the genus c7omp8othlllpis, lacks the yellow characteristic of Setophaga; this basal portion of the outer rectices is dull white instead. The color pattern of the upper parts is distinctly that of the Parula Warbler, and while duller in hue, the olive green of the mantle is in contrast to the color of the lower back. On the other band, the under parts suggest the Redstart, the throat being dull white rather than yellow, with the median portion of the breast tinged with light salmon.
Food: There has been no comprehensive study made of the food of the redstart based on an examlnation of the contents of a representative number of stomachs, but many field observations clearly indicate that it is strictly insectivorous in its food-eating habits. The insects it eats are extremely varied; it gleans them in the form of eggs, larvae, pupae, and adults from the trunks, limbs, twigs, and leaves of trees; and it is most adept in capturing insects in the air. There are few small forest insects, whatever their stage of development, that escape this active and resourceful warbler, so busy is it in pursuit of its prey. Even the caterpillars that escape from other slower warblers by hanging by their silken fibers are readily snatched up by this aerial acrobat. It feeds on insects injurious to deciduous trees and in Maine it frequents the coniferous forests to supplement its food supply. I have frequently seen it snap up flies and tiny midges, mosquitoes, and black flies, all of which, though not injurious to vegetation, are most annoying to human beings. It feeds upon a great variety of caterpillars including the smaller hairy types. It may be seen in orchards, where various observers have noted its fondness for cankerworms, and it has also been seen to feed on brown-tail and gipsy moths. Forbush (1907) saw one redstart eat 31 gipsy moth larvae before it left the clump of willows in which he was able to watch it at close range. At another time he saw a redstart take 11 brown-tail larvae from an apple tree in the course of 5 minutes.
W. L. MeAtee (1926) in his study of the relation of birds to woodlots in New York State writes: “Rations for the Redstart consist entirely of insects, spiders, and daddy-long-legs. Beetles, including flea beetles, leaf beetles, and round-headed wood borers; caterpillars and moths; and such true bugs as spittle insects, tree hoppers, and leaf hoppers are commonly taken. The Redstart devours also some hymenoptera, mayflies, and diptera including craneflies. Busy at all times in the pursuit of insect prey, the Redstart in the long run must account for vast numbers of the forms injurious to trees.”
While the vast majority of the insects eaten by the redstart are harmful to man’s interest it must be admitted that it does feed on a few beneficial insects such as the parasitic Hymenoptera and Diptera as well as an occasional ladybird beetle which are classed as useful.
Although the redstart is preeminently an insect destroyer it has been known to eat berries and seeds on rare occasions. Aretas A. Saunders (1938) reports that in Allegany State Park, N. Y., they feed on shadbush berries; George A. Petrides (1943) reported them feeding on the berries of Berberi8 bealei; and A. H. Howell (1932) states that in addition to its usual food, while the birds are migrating through Florida, it feeds on magnolia seeds as well as on spiders, plant lice, and scale insects.
Alexander Wetmore (1916) reported on his examination of the food found in 13 stomachs of redstarts, which he collected in Puerto Rico during the months of December, January, February, and April, as follows:
In these animal food amounts to 100 per cent. Small lantern flies (Fuigoridae) occur nine times and form 37.23 per cent of the total. Longicorn beetles make up 0.57 per cent, snout beetles 1.5 per cent and miscellaneous species 2.54 per cent. All are classed as injurious species save a single ladybird beetle, so small that It is lost in the bulk of the others. Moth remains amount to 11.75 per cent, while no caterpillars were eaten. Hymenoptera remains (6.67 percent) were found In five stomachs. Approximately two-thirds of these small species, probably of parasitic habit. One that parasitizes ants was definitely identified as a species of Kapala. Diptera (39.24 per cent) were present in eight of the stomachs examined. One bird had eaten a spider, and another insect eggs, both amounting to Only 5 per cent.
Though present only in winter, this small warbler is a bird of economic importance. It destroys thousands of lantern flies, abundant In the trees and shrubbery, as well as many beetles, moths, and flies. A very small percentage of Its food is taken from beneficial species of insects, the remainder being entirely injurious. This bird is entirely insectivorous and does not feed upon any of the abundant wild berries, a fact which increases Its importance, as It destroys proportionately more insects than do the more or less vegetarian resident species of similar habit.
Small larvae and tiny insects are fed to the young by the adults during the first days of nest life but as the young become older and their demands for food correspondingly greater they are fed larger insects such as adult moths, beetles, locusts, and craneflies. Bernard W. Baker (1944) in observations made of nesting redstarts in northern Michigan writes: “Various kinds of insects and larvae were fed to the young: Mayfly (Ephemera) ,Rosy Mapl~ Moth larvae (Dryocampa rubienda), Rouse Fly (Musca do~nestica), and many others I could not identify. During a Mayfly hatch 90 per cent of the insects brought to the nests were Mayflies. It was not unusual for the male to bring in two or three Mayflies and feed two young on one trip. On one occasion a male brought four Mayflies at once and fed three young.”
The young are usually fed from 4 to 15 times during the course of an hour. At one nest under observation the young were fed by the female 28 times and by the male 22 times during the course of 9 hours. The feeding intervals varied from 1 to 25 minutes, with an average of about 10 to 11 minutes between feedings. Ira N. Gabrielson (1922) who observed a nest of young at Marshalitown, Iowa, states that during the course of ~’/2 hours the nestlings had 30 feedings, 10 of which were by the female and 20 by the male. During these feedings he recognized 22 winged insects, 16 larvae, 1 fly, and 1 spider, but in some cases he was unable to determine the character of the food.
It is obvious that the redstart can be classed as a bird very useful to man’s interests because of its destruction of many harmful insects.
Voice: The song of the redstart is not especially pleasing nor is there any outstanding feature of the song except its extreme variation and the different versions in which it occurs. It is a short monotonous weecliy type and of a high-pitched sibilant quality. I have often been mystified by the vagaries of this versatile singer. The beginner may at first confuse the song with that of the yellow warbler, which it resembles, but the latter is longer and has a different ending. The fundamental difference between the two is a tendency of the song of the yellow warbler to accelerate while that of the redstart retards. Sometimes the redstart will emit a series of notes that are weak and buzzy and suggest the parula’s song, and again they will be like the shrill notes of the blackpoll or the loud wheeze of the black-throated blue warbler.
Aretas A. Saunders has sent us his interpretation and analysis of the redstart’s songs, as follows: “The song of the redstart is not loud, but certain notes are sometimes strongly accented. In 59 records, the songs contain from 4 to 11 notes, averaging about 7. They occupy from 3/5 to 12/s seconds.
“The song varies in pitch from G”’ to E””. Individual songs vary from no change in pitch to a change of three tones. Three of my records are all on the same pitch. Four change pitch only a half tone. Only one has a range of three tones. The average range is just about one and a half tones.
“Songs begin with a series of simple notes or of 2-note phrases. The former sounding like t8cet , tseet, tseet and the latter tseeta, tseeta, Iseeta. Songs may end in three different ways; the first just as it began, without change to the end; the second with a single, strongly accented, high er-pitched note; and the third by a downward slur, strongly accented at the beginning. These two latter forms may be represented as tsita, tsita, tsita, tseet and tsd, tsit, tsit, tsit, tseeo. Of my records, 15 are of the first form, 18 of the second, and 26 of the third.
“These three different songs may all be sung by one individual and there is nothing seasonal about their use. The bird frequently alternates two or more songs, sometimes regularly, sometimes one is sung three or four times as often as the other. One bird sang four different songs one after another, but the order varied with each repetition. So far as I know, this is the only warbler that sings different songs in alternation.
“The redstart sings from the time of its arrival until the middle of July. The average date of the last song, based on 14 years of observation in Allegany State Park, is July 14. The earliest is July 2 (1927) and the latest July 25 (1937) .”
In northern New York, A. Sidney Hyde (1939) states that no songs of the redstart are heard in late July but that songs were regularly heard from August 1 until September 2. Several birds were singing with almost full springtime vigor at Irondequoit Bay on August 20.
Eugene P. Bicknell (1884), in writing of the late singing of the redstart, says:
In some years I have found this species songless soon after the beginning of July. In seasons when it thus early becomes silent singing is resumed in the first part of August, and continues for two or three weeks. But the period of July silence is Inconstant, and sometimes singing is little interrupted through the month. When this Is the case singing seems to cease finally at the end of the month or early In August, and is followed by no supplementary song period. * * * My dates of last songs are limited by the third week of August, except in one exceptional Instance when one of the birds was heard to sing on September 5.
A number of observers have reported seeing the female sing, the song being about the same in quality and length as that of the male.
It has been suggested by critics that these reports were based on seeing the first-year male, which has a plumage similar to that of the female and could be easily mistaken for a female. The male breeds the first year in this plumage and this adds to the confusion. However, it has been definitely proved that the females do sing at times. J. L. Baillie, Jr. (1926) collected a female at Lowbush, Lake Abitibi, Ontario, on June 3, 1925, which was singing one of the usual songs of the male, including the usual 8wee-a, swee-a notes. The specimen was carefully sexed and was found to have enlarged ovaries; hence, no mistake was possible. On June 22, F. W. Braund (Braund and McCullah, 1940) heard what appeared to be a singing male on Anticosti island. When seen it was thought to be a young male in immature plumage in full song, but after it was collected and sexed it proved to be an adult female. These and other similar cases indicate that some females sing the full song of the males, although it is unusual for them to do so.
Albert R. Brand (1938) has determined the vibration frequencies of the songs of many passerine birds from motion-picture film recordings. The highest note in the song of the redstart had a frequency of 7,300 vibrations per second, the lowest 4,400, and the approximate mean of 6,200 vibrations per second. It is of interest to compare the mean of the redstart with that of the blackpoll warbler, which has a mean of 8,900, whereas the mean of the yellow-breasted chat is only 2,600 vibrations per second.
Horace W. Wright (1912) has observed the early morning awakening of birds at Jefferson Highlands in the White Mountains of New Hampshire. Of the redstart he writes: “If the Ovenbird is excepted, the Redstart introduces the warbler singing. All warbler song is delayed on the average until 3.29, when the Ovenbird begins to sing. The Redstart based on ten records, averages to sing seventeen minutes later, or at 3.46 o’clock. It has been heard once as early as 3.26, but on three other occasions was first heard at 3.55, 3.56, and 3.58 respectively. Two and sometimes three birds sing within hearing. On June 28, 1911, the first bird began to sing at 3.41, the second was heard at 3.49, the third at 3.52, and the three continued singing much of the time up to 5.15 o’clock.” He states further that the Redstart was one of only three warblers which were beard to sing after sunset., which at the time the records were taken was at 7.30 p.m.
Enemies: The redstart’s habit of nesting in rather tall slender trees, usually at a considerable distance from the ground, frees it from most of the enemies to which ground-nesting birds are subject. However, I know of one nest of the redstart, built in a shrub about 4 feet above the ground, that was destroyed by a cat, and of another, also located in a shrub near a house, that was quite probably destroyed by one. However, since the vast majority of redstarts build in locations remote from human habitations, cats are not an important factor in affecting the redstart population as a whole.
John and James M. Macoun (1909) cite a case of a redstart’s nest, built in an exposed position, that was presumably destroyed by an olive-sided flycatcher that had a nest on an overhanging branch a few rods away.
C. H. Morrell (1899) states that two redstart nests which he observed were deserted after the eggs had been laid because caterpillars had taken possession of the nesting tree and had completely overrun t.he nests. E. H. Forbush (1907) in connection with a gipsy moth infestation at Medford, Mass., writes: “There was a nest of the American redstart and the tree had been stripped of leaves by the caterpillars. There were four young in the nest. I saw the old birds take but one very small gipsy moth caterpillar to the young, but they would pick the large ones off the nest and drop them to the ground very often. There were no pupae near the nest that I could see.” Mr. Forbush does not state whether the redstarts succeeded in winning their fight against this caterpillar infestation.
Louis Sturm (1945) says that one nest that he observed “was robbed by a small fox snake (Elaphe vulpina gloydi) which had swallowed all three eggs when discovered coiled in the nest.”
The redstart, like most birds, is host to a number of external parasites, of which Harold S. Peters (1936) has identified the three species of lice Menacanthus sp., Myrsidea incerta (Kellogg), and Philopterus sulflavescens (Geoffroy) and the tick Haemaphysalis leporia-pahistris Packard.
Perhaps the greatest menace to the redstart is the parasitic cowbird, of which this warbler is one of the commonest victims. According to Herbert Friedmarin (1929) at Ithaca, N. Y., the redstart was the most imposed upon species. Out of 34 nests he found 23 of them contained one or more of the parasitic eggs and it was not uncommon to find sets composed of but one or two of the rightful eggs, the rest being the cowbird’s.
Occasionally the redstart builds a new floor over the cowbird’s eggs, as is often done by the yellow warbler. This procedure is especially likely to take place if the cowbird succeeds in laying its eggs before the redstart’s eggs are present. More often the redstart does not seem to be annoyed by the strange eggs but goes on laying its own. It is unusual for the redstart to desert its nest because of the presence of cowbird’s eggs.
A nest was found on June 9, 1922, with two young cowbirds 5 days old, completely filling and covering the nest. In the bottom beneath the young parasites were four addled eggs of the warbler. If some of the rightful eggs hatch, the young are usually starved or suffocated by the young cowbirds, although nests are occasionally found in which one or more of the young survive along with the interloper.
In discussing the effect of this parasitism on the redstart Friedmann adds: “When a cowbird is raised by a one-brooded species such as the redstart it represents the total product of its pair of foster-parents for the year. The loss here is a very decided one if we consider the food consumed by one cowbird and that which would have been eaten by four Redstarts. However, judging from the constancy of the numerical status of the Redstart from year to year it seems as though three of the four young would succumb anyway to various dangers before the next year. Also we must remember that the Redstart is in many places more abundant than the cowbird.”
Joseph J. Hickey (1940) writes: “Males were silent in the presence of female Cowbirds, but females reacted with sharp hisses, a rapid snapping of the bill and much spreading of the tail.” Friedmann (1929) writes: “On July 2, 1921, a young Cowbird, full grown in size and fully fledged was seen following a Redstart and begging for food from it. The Redstart paid absolutely no attention to it although several times the two were very close together. All this time the Redstart was busy gathering food and when it bad as much as it could carry it flew off and the young Cowbird did not follow.”
Dayton Stoner (1932) gives an account of an interesting experience of the behavior of redstarts as follows: “I saw one female carrying food for young. Another female was seen feeding a young cowbird that was out of the nest and able to fly, while an adult female cowbird sat on a limb nearby and apparently watched the proceedings. The female cowbird did not offer the young one any food, but after the latter’s wants had been satisfied in some measure by the diminutive redstart, she moved close to the young one and at least appeared to be solicitous of its welfare. But any maternal solicitude involving real care of the young was utterly foreign to this parasitic bird.”
Fall: The redstarts start migrating from the most northern sections of their nesting range in August but it is well into September before the last individuals leave these outposts. The individuals breeding in British Columbia near the Pacific Ocean do not follow the Pacific flyway but retrace their path across the mountains to the interior and leave the United States between Texas and Florida, their point of entry. In recent years an increasing number of redstarts have nested in Washington and Oregon, and there are many records of occurrence, especially during the time of migration, throughout California and southward. This may mean a building up of a tendency of the individuals nesting in the northwest to use the Pacific flyway, which in the course of a few hundred years may become a regular migration route for these Redstarts.
At Point Pelee, Ontario, according to Taverner and Swales (1908) the bulk of the redstarts pass through during the first week of September but are fairly common during the latter part of the month. In Maine the bulk of the redstarts leave by the middle of September but in southern New England and New York there are many records of redstarts throughout the month of October.
In Ohio and the Middle West there is a distinct movement of redstarts early in August and the peak of the migration takes place from August 20 until September 25. A sharp decrease takes place during the last few days of September, and by October 5 the species disappears except for an occasional straggler.
Thomas D. Burleigh (1944b) in discussing the fall migration on the Mississippi Gulf coast writes: “The first birds to appear are always young of the year, and although a few females are seen early in August, it is not until after the middle of the month that the males are observed. During September the Redstart occurs in its greatest numbers, and there is usually an occasional day after the middle of the month * * * when these warblers are found literally everywhere.” Mr. Burleigh’s latest fall record is November 10, 1937. In Florida the largest flights also occur about the middle of September, and at this time many are reported killed at the lighthouses. Many are seen throughout October and some late fall records are: Pensacola November 19, 1929; Fort Pierce November 2, 1918; Fort Drum November 3, 1888; and Sombrero Key November 4, 1888.
According to F. C. Lincoln (1935), redstarts, evidently the more southern breeders, are seen returning southward on the northern coast of South America just about the time that the earliest of those breeding in the North reach Florida on their way to winter quarters.
Winter: The redstart may be found in the West Indies from August until the following April. In Cuba, according to Thomas Barbour (1923), “the Redstarts are the first sign to the Cubans that the migration is on, and they probably are the very last northern visitor to leave in the spring. They are excessively abundant in thickets and woods everywhere, even in the cities. Except for the Palm Warbler, no bird is more in evidence during the winter than the Candelita: the little flame. The North American observer never would dream. that there could be enough redstaits gathered together from all the bird’s range to make up the hordes which come to Cuba.”
Alexander Wetmore (1916), in his account of the birds of Puerto Rico, writes of the redstart: “These birds frequent the mangroves, forest growths, and coffee plantations, and sometimes shade trees about houses. The greater portion seen were immature birds and females, though occasionally males in full plumage were observed. As always, these birds were very active, searching through the limbs for insects, and expertly catching insects on the wing. On El Yunque, in the dense forests, they were seen up to 1,500 feet altitude, and elsewhere in more open locations were found in the highest elevations. Next to the parula warbler this is the most common of the wintering warblers.” In Haiti and the Dominican Republic, Wetmore and Swales (1931) found that “the redstart frequents shrubbery, groves and forests, where it is a most active flycatcher, pursuing its living prey with dash and vigor among the branches with much display of its brilliantly marked, fan-shaped tail. It ranges from coastal thickets to the summits of the mountains.”
Todd and Carriker (192~),in their account of the birds of the Santa Marta region of Colombia, state that the redstart is “an abundant bird during the winter months in the mountains, but much rarer in the lowlands. Its habits are practically the same as in the north, except that it does not sihg. Simon speaks of finding it in the densest forest in the tops of the highest trees, where it is hard to see.” The earliest fall arrival date reported by the authors was August 24, 1898, and the latest spring date May 1, 1913.
Dr. A. F. Skutch has written us an account of the redstart as he found it wintering in Central America as follows: “First appearing in Central America in mid-August, the redstart spreads rapidly over the lower portions of the whole great isthmus. Although during its periods of migratory flight it is at times met in the highlands, even up to an altitude of 8,000 feet, like the majority of the warblers that breed in the Austral region of North America it prefers the warmth of the lowlands. Once it has settled down in its winter home, it is only rarely found as high as 5,000 feet. At lower altitudes, it is a fairly common winter resident throughout the length and breadth of Central America. Although not rare on the Pacific side, it is more numerous in the Caribbean lowlands. In the northern lowlands of Honduras and Guatemala, it is very much in evidence during the winter months, and may be expected wherever it can find trees in which to forage. Incessantly active, it retains in the Tropics the same sprightly ways that make it a favorite of northern bird-lovers, and as it weaves skillfully through the boughs of the trees in pursuit of insects on the wing, conspicuously displays the bright orange or yellow areas on its sides, wings and tail. Like nearly all of the warblers that winter in the lowlands, it is quite solitary during its sojourn in Central America, never associating with others of its own kind. I have not heard it sing while in the Tropics.
“Like the black-and-white warbler, the redstart begins to withdraw from Central America considerably earlier than many other of the wintering warbiers. As a rule, it disappears from Costa Rica before the end of March. I have April records for only two individuals, one of which, a female, lingered as late as the sixth. In Guatemala, however, it has been reported as late as May 6.”
DISTRIBUTION
Range: North America, south to the West Indies and northern South America.
Breeding range: The redstart breeds north to northern British Columbia (Atlin, Telegraph Creek, and Liard Crossing); Yukon; central western and central southern Mackenzie (Fort Norman, Fort Wrigley, Fort Simpson, and Fort Resolution); central Saskatchewan (Emma Lake and Cumberland House); southern Manitoba (Duck Mountain and Berens Island); central and northeastern Ontario (Kenora, Lao Seul, Rossport, and Moose Factory); central western and southeastern Quebec (probably Rupert House, Mistassini Post, Godbout, Matamek, Mingan, and Anticosti Island); and Newfoundland (Bay of Islands, Twillingate, and Fogo Island). East to Newfoundland (Fogo Island); Nova Scotia; New Brunswick; Maine (Andover and Auburn); New Hampshire (Hollis); Vermont (Newfane); Massachusetts; New York; southeastern Virginia (Dismal Swamp); central North Carolina (Charlotte and Raleigh); and central Georgia (Atlanta, Macon, rarely Americus, and once in Savannah. South to central Georgia (Savannah); central Alabama (Greensboro, Booth, and Seale); central Mississippi (Edwards); northwestern, central and southeastern Louisiana (Mansfield, Natchitoches, Lottie, and Diamond); central and southeastern Oklahoma; central northern Colorado (Central City, Longmont, and Boulder); and central Utah (Provo). West to central Utah (Provo and Parleys Park) ; northeastern Oregon (probably Minan) ; central northern Washington (Winthrop and Chelan) ; and western British Columbia (Hagensborg, Pemberton, and Atlin).
Winter range: The redstart winters north to southern Baja California (Miraflores) ; Veracruz (Presido and Tres Zapotes) ; Yucahin (Chich6n-Itz~) ; Quintana Roo (Holbox Island and Cozumel Island); Cuba (Mariel, Guamo, and Gibara) ; rarely the Bahamas (Green Cay, Andros, New Providence, and Great Inagua); Hispaniola (Fort Libert~, Sousa, and SThchez) ; and Puerto Rico (Mona Island, Mayagiiez, and Culebra Island). East to Puerto Rico (Culebra Island); the Lesser Antilles (Virgin Islands, Barbuda, Antigua, Dominica, Santa Lucia, Grenada, Tobago, and Trinidad); and British Guiana (Mount Roraima and Bartica). South to British Guiana (Bartica); northwestern Brazil (Sierra linen); and Ecuador (Zamora). West to Ecuador (Zamora, Quito, and Esmeraldas); Puebla (Metlatoyuca) and southern Baja California (San J05~ del Cabo and Mirafiores).
The range as outlined is for the two recognized races. The Southern redstart (Setophaga ruticilla r’uticilla) breeds from North Dakota to Maine, south to Georgia, Louisiana, and central and southeastern Oklahoma; while the northern redstart (S. r. tricolora) breeds from northern British Columbia to Nova Scotia, south to central northern Colorado, and west to central northern Washington and northeastern Oregon.
Migration: Late dates of spring departure are: Ecuador: below San Josh, March 15. Venezuela: Rancho Grande near Maracay, Aragua, May 5. Colombia: La Tigrera, Santa Marta region, May 1. Panam~: Gatin, April 28. Costa Rica: El General, April 6. British Honduras: Mountain Cow, April 14. Swan Island: April 19. Guatemala: Chuntugui, May 3. Tabasco: Balancan, May 16. Virgin Islands: Kingshill, St. Croix, April 30. Puerto Rico: Cabo Rojo Light, April 26. haiti: tIe de Ia Gon~ve, May 20. Cuba: Cienfuegos, May 13; Habana, May 11 (average of 8 years, May 4). Bahamas: Nassau and Cay Lobos Light, May 13.
Early dates of spring arrival are: Venezuela: IRancho Grande near Maracay, Aragua, February 27. Florida: Key West, March 6; Fort Myers, March 12; Pensacola, April 4. Alabama: Birmingham, April 1. Georgia: Milledgeville region, March 16; Athens, April 7. South Carolina: Clarendon County, March 24; Marion, April 2. North Carolina: Raleigh, April 2 (a.verage of 27 years, April 12). Virginia: Charlottesville, April 13 (average, April 22). West Virginia: Kayford, April 10; French Creek, April 22 (average of 17 years, April 29). District of Columbia: Washington, April 16 (average of 53 years, April 22). Maryland: Largo, Prince Georges County, April 15. Delaware: Kent and Sussex Counties, February 28 (average of 22 years, March 20). Pennsylvania: Berwyn, April 3; Pittsburgh, April 21; Renovo, April 29 (average of 25 years, May 4). New Jersey: Bridgeton, April 23. New York: Bronx County, April 24; Ithaca, April 29. Connecticut: Hartford, April 22. Rhode Island: Providence, April 29. Massachusetts: Springfield, April 28. Vermont: Bennington, Wells River, and Bakersfield, May 4. New Hampshire: South Hooksett, May 2. Maine: Bowdoinham, May 3. Quebec: Montreal, May 4. New Brunswick: St. Stephen and Bathurst, May 12. Nova Scotia: Wolfville, May 3. Prince Edward Island: North Bedeque, May 24. Newfoundland: Tompkins, May 29. Louisiana: New Orleans and Grand Isle, April 7. Mississippi: Edwards, March 23. Arkansas: Helena, March 31. Tennessee-Nashville and Chattanooga, April 12 (average of 10 years at Nashville, April 20). Kentucky: Bowling Green, April 4. Missouri: southeastern Missouri, April 11. Illinois: Addison, April 9 (average for Chicago region, May 7). Indiana: Bloomington, April 12. Ohio: Oberlin, April 7; Buckeye Lake, April 25 (average May 2). Michigan: Ann Arbor, April 25. Ontario-London, April 30; Ottawa, May 3 (average of 17 years, May 10). Iowa: Wall Lake, April 21. Wisconsin: Milwaukee, March 20; Berlin, April 28. Mmnesota: Lanesboro and Red Wing, May 2 (aver age of 45 years for southern Minnesota, May 10). Texas: Port Arthur, March 23. Oklahoma: Tulsa, April 10. Kansas: Manhattan, April 22. Nebraska: Red Cloud, April 5, April 17 (average of 20 years, May 12). South Dakota: Yankton, May 3. North Dakota: Cass County, May 7 (average, May 14). Manitoba: Brandon, May 8; Aweme, May 11 (average, May 15). Saskatchewan: Indian Head, May 12. Mackenzie: Fort Simpson, May 20. Arizona: Yuma, April 30. Colorado-Fort Morgan, May 5. Utah: Ogden, May 22. Wyoming: Careyhurst, April 30; average of 8 years for southern Wyoming, May 17. IdahoRatlidrum, May 11. Montana: Billings, May 12. Alberta: Camrose, May 9. Nevada: Las Vegas, May 21. Washington: Cheney, May 19. British Columbia: Okanagan Landing, May 20. Alaska: Wrangell, June 9.
Late dates of fall departure are: British Columbia: Shuswap Falls, September 22. California: Verdugo, Los Angeles County, November 23; Pasadena, December 27. Alberta: Glenevis, September 22. Montana: Great Falls, October 3. Idaho: Moscow, September 4. Wyoming: Laramie, September 12 (average of 5 years, September 4). Utah: Uinta Basin, September 20. Colorado-Yuma, October 22. Arizona: Imperial National Wildlife Refuge, September 22. Saskatchewan: Wiseton, October 2. Manitoba: Aweme, September 28 (average, September 18) ; Winnipeg, October 26. North Dakota: Cass County, September 23 (average, September 17); Rice Lake, September 25; Fargo, November 1. South Dakota: Faulkton, October 15. Nebraska: Omaha, September 10. Kansas: Harper, September 21. Oklahoma: Tulsa, September 25. Texas: Cove, November 6, December 26. Minnesota: Virginia and Faribault, October 12 (average of 14 years for southern Minnesota, September 23). Wisconsin: La Crosse, October. 11. lowa: Sigourney, October 12. Ontario: Ottawa, October 3; Point Pelee, October 15; Toronto, November 28. Michigan: Detroit, October 11. Ohio: central Ohio, October 27 (average, October 6); Hillsboro, November 1. Indiana: Lake or Porter County, October 11; Carlisle, October 29. Illinois: Chicago, October 31 (average, October 8). Missouri: Keokuk, October 20. Kentucky: Danville, October 23. Tennessee: Elizabethton, October 19. Mississippi: Gulf coast, November 10. Louisiana: Baton Rouge region, November 7. Newfoundland: Stephenville, September 21. Prince Edward Island: North River, September 7. Nova Scotia: Yarmouth, August 31. New Brunswick: Saint John, September 25. Quebec: Montreal, October 6. Maine: Jefferson, October 11. New Hampshire: New Hampton, October 9. Vermont: Wells River, October 17; Bennington, November 11. Massachusetts: Cambridge, November 7; Belmont, November 16. Rhode Island: Little Compton, October 22. Connecticut: West Hartford, October 24; Windsor, November 25. New York: Ithaca, October 18; New York City, November22 and 23, December 27. New Jersey: Englewood region, October 19. Pennsylvania: Sewickley, November 28; Lakemont, Blair County, December 25. Delaware-Kent and Sussex Counties, October 16 (average, September 10). Maryland: Patuxent Wildlife Research Refuge, October 16; White Marsh, December 6. District of Columbia: Washington, November 16 (average of 33 dates, September 20). West Virginia: Bluefield, October 12. Virginia: Cape Henry, November 28 (average, September 28). North Carolina: Raleigh, October 13 (average of 12 years, October 3); Statesville, October 16. South Carolina: Clarendon County, October 21. Georgia: Athens, October 27. Alabama: Birmingham, October 22, December 17. Florida: Pensacola, November 19. Bahamas: Cay Lobos Light, October 20.
Early dates of fall arrival are: British Columbia: Francois Lake, August 5. California: Monterey Park, August 30. Idaho: Jerome, July 7; Jensen, August 20. Colorado: Yuma, August 26. Arizona-Oracle, August 12. New Mexico-Kingston, August 24. North Dakota: Cass County, August 20 (average, August 27). South Dakota: Faulkton, August 15. Nebraska: Red Cloud, August 21. Oklahoma: Norman, August 30. Texas: Austin region, August 17; Brownsville, August 31. lowa: Osage, August 11. Ohio: Buckeye Lake, July 23 (average, August 4). Illinois: La Grange, August 2; Chicago, August 9 (average, August 14). Mississippi: Gulfport, July 16. Maine-Bailey Island, July 30. Massachusetts: Ghatham, August 27. New York: New York City, August 2. New Jersey: Milltown, August 6. Maryland: Patuxent Wildlife Research Refuge, August 12. Virginia: Cape Henry, September 3 (average, September 10). North Carolina: Washington, August 10. South Carolina: Clarendon County, July 18. Georgia: Athens, July 22. Alabama: Leighton and Birmingham, August 23. Florida: Alligator Point, Franklin County, July 19; Fort Myers, July 28. Cuba: Cienfuegos, August 6. Jamaica: St. Elizabeth, August 10. Dominican Republic: Constanza, September 22. Puerto Rico: Cabo Rojo, September 17. Barbuda, August 10. Guatemala: Puerto Barrios, August 21. Salvador: Lake Olomega, August 27. honduras: near Tela, August 19. Costa Rica: El Hogar, August 16. PanamA: Almirante Bay region, August 27. Colombia: Santa Marta region, August 24. Venezuela: Rancho Grande, near Maracay, Aragua, August 31. Ecuador: Esmeraldas, October 18.
Egg dates: Illinois: 32 records, May 25 to July 21; 16 records, May 29 to June 6, indicating the height of the season.
Michigan: 14 records, May 25 to July 12; 7 records, May 30 to June 8. Maine: 20 records, June 5 to 24; 11 records, June 10 to 17.
Massachusetts: 64 records, May 22 to June 30; 40 records, May 30 to June 6.
Nova Scotia: 13 records, June 3 to 25; 7 records, June 15 to 18..
Pennsylvania: 7 records, June 2 to 16 (Harris).
NORTHERN AMERICAN REDSTART
SETOPHAGA RUTICILLA TRICOLORA (Muller)
CONTRIBUTED BY JAMES LEE PETERS
HABITS
The name Motacifla tricolora was applied by Milller (1776) to a colored figure of a redstart in Daubenton’s Planches Enlumin~es (1765, p1. 391, fig. 2), Muller apparently not recognizing that this figure actually represented the bird named Motacilla ruticilla by Linnaeus in 1758. Muller’s name remained in the synonymy of ruticilla until resurrected by Oberholser (1938) who applied it to a western race of redstart which he characterized as differing “from the eastern form in smaller size, smaller orange or yellow wing spot; in the female also paler, more grayish, less conspicuously olivaceous, upper surface.”
As breeding range he assigned northern British Columbia, Mackenzie, and central Manitoba, south to Oregon, northern Utah, and Wisconsin; he believed that it migrated through the greater part of the United States and wintered in South America to Ecuador and French Guiana. Wetmore (1949) found that the characters given by Oberholser for distinguishing S. r. tricolora from S. r. ruticilla did not hold, and the range of the former was more extensive than supposed. As a result of his investigations Wetmore found that the size of the wing patch was variable and hence not a subspecific character; in fact he could find no constant differences between the adult males of the two races, but found that females and immature males of tricolora differ from ruticilla in being “somewhat darker above, washed with duller green in immature dress.”
The breeding range for tricolora given by Wetmore extends from Yukon, Mackenzie, Saskatchewan, central Ontario, and Quebec to Newfoundland, south to west-central Washington, northern Utah, Montana, northern Maine, New Brunswick, and Nova Scotia.
Both forms appear to migrate and winter together.


